July 2025 Launch: Technical Standard to Confirm Natural Health Product Effectiveness
Toronto, Canada — July 2025 – The Association of Certified Professional Nutritionists (ACPN) proudly announces the official publication of the Technical Standard for Evidence-based Efficacy System of Natural Health Products (自然健康产品的循证功效体系技术标准). This landmark document establishes the first comprehensive, multi-platform technical guideline designed to confirm and standardize the efficacy of natural health products. This initiative represents a significant step forward for the nutrition and natural health industry in Canada and beyond, aligning with ACPN’s mission to promote science-based health practices.
The Technical Standard was developed through collaboration between leading scientific organizations, including Keypoint Boston LLC, the Royal Academy of Nutrition Science, National Laboratory of Nutrition Science, and the International Quality & Standard Society, with sponsorship and co-sponsorship by ACPN Canada, ACPN USA, M2 CPN Clinic, Markham CPN Clinic, General Nutrition Group Inc., and other partners. This broad international partnership ensures the document reflects global best practices and is applicable across regulatory contexts.
This new standard is now being distributed to natural health product producers, research institutions, and member clinics across ACPN’s network. ACPN formally recognizes this procedure as the authoritative method for confirming the efficacy of natural health products and will encourage its adoption as part of its professional practice guidelines. This will provide consumers, practitioners, and regulatory authorities with more reliable evidence of product benefits, increasing trust and transparency in the marketplace.
The 78-page document covers the full scope of evidence-based efficacy evaluation, from objectives and scope to data analysis and reporting . Key sections include detailed preparation guidelines for zebrafish, mammalian, and human study platforms, strict adherence to international ethical requirements such as the Declaration of Helsinki, and robust statistical approaches for data processing and validity determination.

The book provides step-by-step experimental procedures: zebrafish rapid screening for functional efficacy, mammalian model confirmation with CMA-compliant safety testing, and human dietary intervention trials for clinical endpoint validation. It also specifies criteria for informed consent, participant recruitment, and sample size determination, ensuring that human trials are ethically conducted and scientifically robust .


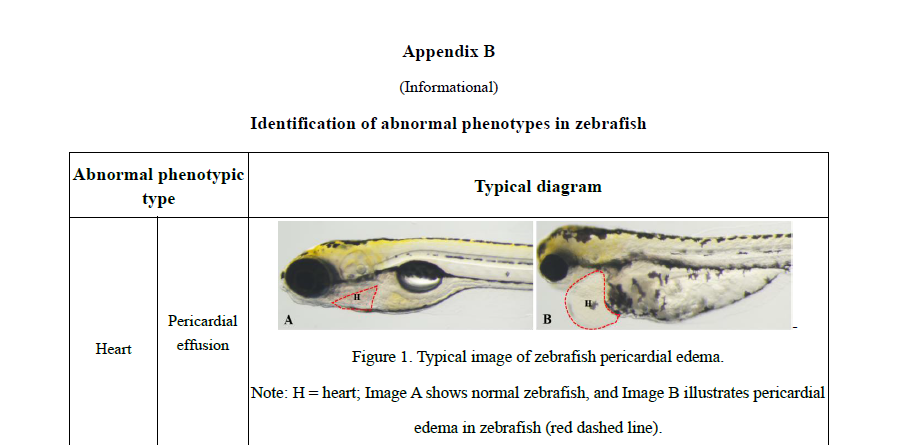
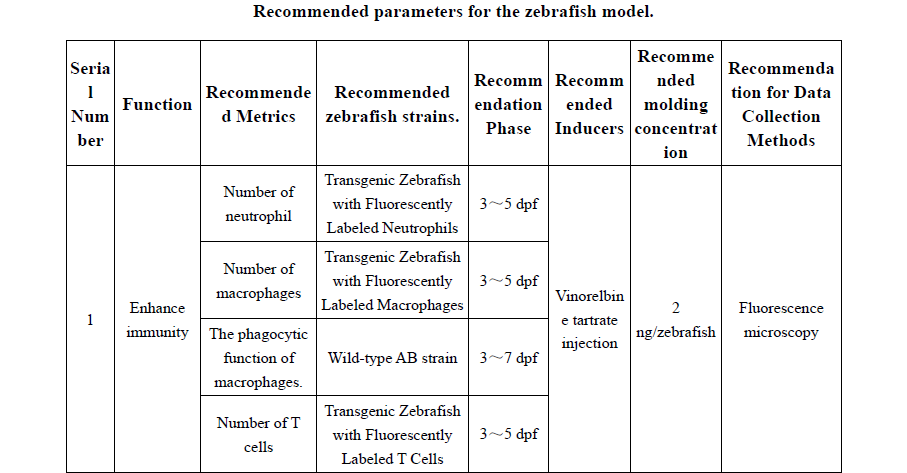
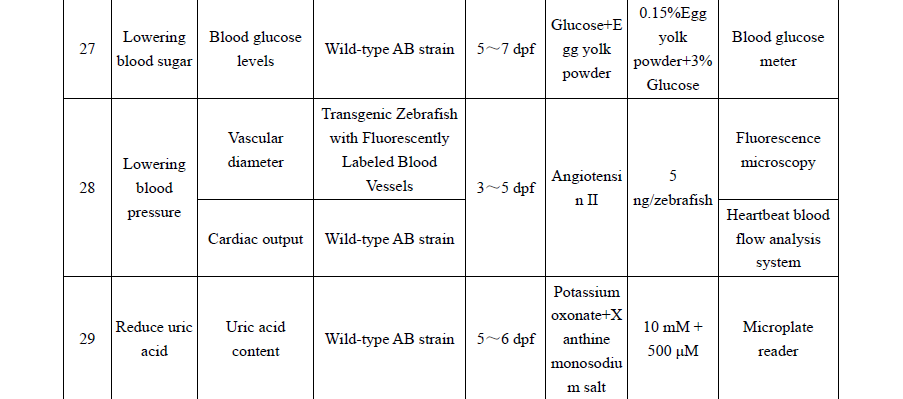
Practical appendices expand the book’s usability, with recommended zebrafish models for 30+ functional outcomes (e.g., immunity enhancement, antioxidant capacity, sleep improvement, lipid-lowering effects), guidance on mammalian pathological models, and phenotypic identification criteria. These tools allow researchers and manufacturers to apply standardized methodologies and improve reproducibility and comparability of results across laboratories .
By releasing this Technical Standard, ACPN reaffirms its commitment to promoting evidence-based nutrition science and supporting innovation in natural health products. ACPN encourages producers, clinical researchers, and healthcare professionals to adopt these guidelines to ensure the safety, quality, and proven efficacy of products reaching consumers. The distribution of this standard is a milestone toward establishing a transparent and credible system for validating natural health product claims, ultimately benefiting both the industry and public health.